|
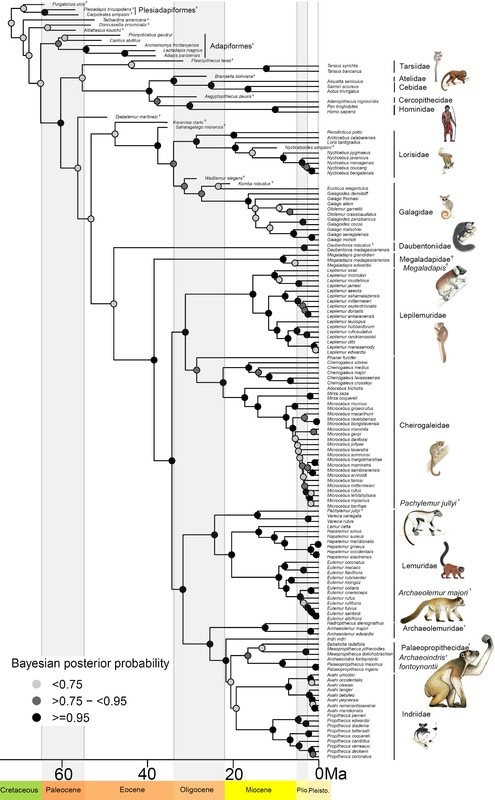
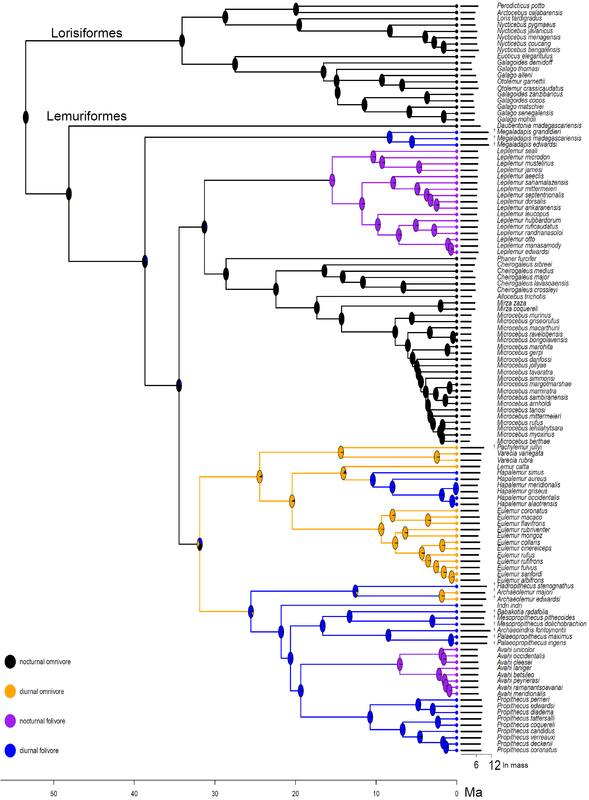
In a new article available online at the Royal Society Open Science, I tested several predictions of adaptive radiation theory for lemurs of Madagascar. This charismatic group has long been suggested to be the result of adaptive radiation, but my results suggest the long evolutionary history of lemurs may not fit all the predictions. Adaptive radiation is a key concept in evolutionary biology, with a fantastic diversity of species arising to fill every conceivable niche. Famous examples include Darwin’s finches of the Galapagos, lizards of the Caribbean, and cichlid fishes in Africa. Madagascar, an island nation off the coast of Africa, is a biodiversity hotspot, with thousands of species found nowhere else on earth. The primates – lemurs – have long been suggested to be an example of adaptive radiation, with around 100 species alive today that have unique and diverse niches. The theory of adaptive radiation makes several explicit predictions, which have not yet been tested for lemurs. For example, evolution of morphological adaptations to fill unique niches is an important part of adaptive radiation theory. Do lemurs fit these predictions? In a new article accepted in the Royal Society Open Science, I tested multiple predictions about the rate that species diversified, and how their body size adapted to different niches of diet and activity pattern. I also tested the possibility that a mass extinction event in the past, 34 million years ago, may have changed the rate of speciation and extinction. While theory predicts that speciation rates should be highest early in the evolution of an adaptive radiation and slow down towards the present, lemur speciation rates were either constant through time, or may have even increased slightly over time. The rate of body size evolution fit predictions better than speciation rates; the major groups of lemurs seem to have differentiated into unique niches with different optimal body sizes. Lemurs that eat leaves and are active during the day have larger body size than day-active fruit eating lemurs. There are three unique groups of small-bodied lemurs that have independently evolved adaptations for eating leaves. Additionally, there are small nocturnal species adapted for eating fruits, nectar, and insects. This diversity of niches and associated body size evolution suggests that lemurs may have evolved via adaptive radiation, despite the contrary results from speciation rates. While there was no evidence that a mass extinction event disrupted speciation dynamics in the deep past, 17 species of giant lemurs have gone extinct within the last 2,000 years, not long after the arrival of humans around 4,000 years ago. The giant lemurs were unlike most alive today, with one species as large as a silverback gorilla. The loss of these unique species represents a significant change in Madagascar ecology and evolutionary history. This study provided explicit tests of the adaptive radiation theory for lemurs, with mixed results suggesting that body size and niche evolution fit expectations, but speciation rates may tell a different tale. Lemur phenotypes - or their morphology, ecology, and behavior - are so diverse, they fit the prediction of adaptive radiation. While some species are small, nocturnal omnivores, including the smallest primates, the mouse lemurs, other lemurs are larger, diurnal and eat leaves. Some species are even specialized to eat bamboo with enough cyanide to kill people! The largest living lemur, the 6-10kg (12-20lbs) Indri, is tiny compared to the giant lemurs that went extinct just 2,000 years ago, which were as big as gorillas - up to 150kg, or 300lbs! Phylogeny of lemurs and other primates, illustrating living and extinct species. Figure from Herrera and Davalos 2016, Syst. Biol. Illustrations courtesy of S. Nash, used with permission. Strepsirrhine phylogeny illustrating the distribution of body mass (black bars next to tips, scale at the bottom, natural log transformed) and adaptive niches of diet and activity pattern (colored circles next to tips). The evolution of niches was estimated at internal nodes (colored pie charts on nodes representing the proportional probability of each state) and the best estimate at each node was assigned to the descendant lineages (colored branches). This tree was used to test how body mass evolved at different rates in different niches.
0 Comments
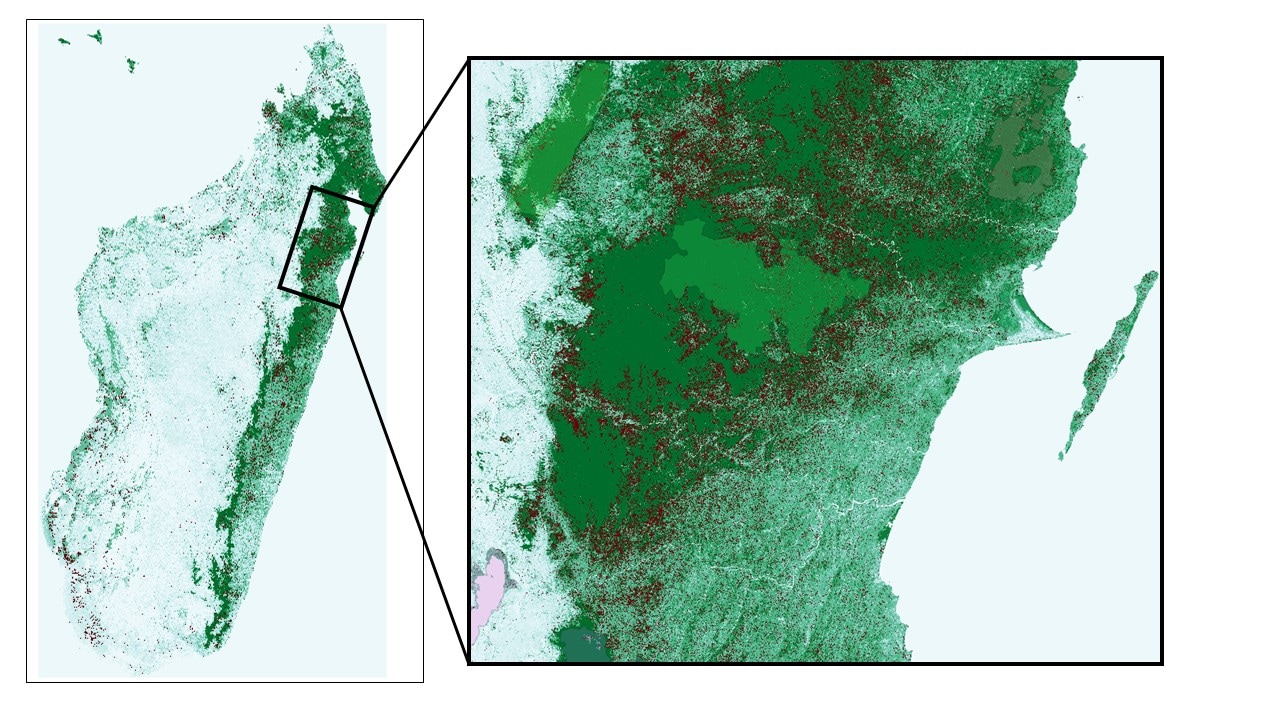
My latest paper on lemur conservation priorities just came out in the journal of Biological Conservation. In it, I discuss a new priority scheme for 100 protected areas that have high lemur diversity in terms of threatened species, functional traits of species, their evolutionary history, as well as the quality and threat to their habitat. This multidimensional approach allowed me to rank protected areas based on a diversity of measures. Biodiversity worldwide is threatened with extinction due to human activities. Primates, the group including monkeys, apes, and people, are vulnerable to habitat loss, hunting, and future climate change. 44% of primate species are considered threatened by the International Union for the Conservation of Nature and Natural Resources (IUCN). The lemurs of Madagascar, found nowhere else, are the most endangered primate group, with 93% threatened due rapid habitat loss, as well as hunting in some areas. There has been a tremendous effort to conserve lemurs and the habitats they thrive in, including the creation of over 100 parks and reserves (protected areas, PAs) to protect montane rainforests, tropical dry forest, and arid spiny forest. Madagascar is an impoverished nation, however, with limited funds to maintain PAs. In 2013, conservation priorities for PAs were set based on the number of species in each PA and how endangered they are in the Lemur Action Plan. In a new study, I propose a new priority ranking for 100 PAs based on their lemur diversity and habitat quality. Assessing diversity is difficult, though – how should we calculate the value of a PA for lemurs? I quantified diversity in terms of 1) the number of species, 2) how endangered those species are, 3) how diverse the functional traits of species are, 4) how much evolutionary history species represent, and 5) how diverse and productive the habitats are. To measure the level of anthropogenic threat to these PAs, I calculated the rates of deforestation using data from Hansen et al. 2013 (data found here). I found that the priority ranking of PAs differed depending on the measure used, leading me to create a summary metric that captures the importance of each PA on multiple levels of diversity and threat. Surprisingly, I found that deforestation inside of PAs was high; up to 31% forest loss between the years 2000 and 2014, totaling ~3,000 km2, which is 50 times the size of Manhattan. The highest deforestation occurred in PAs with high species richness and functional trait diversity. I ranked PAs to suggest the priorities for more intensive future conservation efforts. The top PA is a corridor in the southeast that links the Fandriana National Park to the Marolambo forest south of it, and has only recently been declared. Corridor forests are especially important as a means for individuals to disperse among populations and aid gene flow, as well as a means of repopulation if some populations suffer declines. Based on phylogenetic diversity - the evolutionary distance separating the species in the park - some internationally recognized PAs were found to be priorities, such as one UNESCO world heritage site (Tsingy de Bemaraha). There are also several highly threatened environments with high functional diversity, especially in the arid southwest like Beza Mahafaly. With these new priority rankings, scientists can collaborate to focus their efforts on the most threatened and diverse PAs. Efforts should also be maintained in successful PAs, which have had limited deforestation. These sites have created partnerships with local stakeholders to collectively protected natural habitats, such as Ranomafana and Analamazaotra. The full list of PAs, and their ranks based on each metric can be downloaded here. While there are many impediments to conservation in Madagascar, especially high deforestation for agriculture, there are many success stories which provide a framework for conserving species in the future. Much remains to be learned about conservation priorities in Madagascar. It is especially important to quantify the importance of these PAs for the people of Madagascar. PAs are important at the national level because of the revenue generated by tourism. They significantly impact the lives of local populations surrounding them, who depend on the land for subsistence, like farming and building materials, as well as for ecosystem services such as clean water. Parks generate revenue for local populations through tourism and research, but often the economic loss of restricted access to land is not offset. Lastly, some forests have spiritual value, because some ethnic groups bury their dead in tombs within forests. Therefore, nature conservation must also determine the value of PAs for people. Another way this study can be improved is to enumerate these diversity data for multiple taxonomic groups. These data are accumulating with many field projects collecting data on diverse groups such as birds, small mammals, bats, amphibians, reptiles, and plants. Combining these new occurrence records for diverse groups will allow a more multifaceted approach to conservation in Madagascar. To summarize, this new paper reports conservation priorities based on the sum of lemur diversity, habitat quality, and deforestation. Rapid loss of habitat is threatening important biological systems that also provide crucial services for people. I hope that the results of this study will be well received by the community and can help guide future conservation efforts. Below are figures illustrating the extant of forest in the year 2000 (green gradient) and the forest loss is in red. I also zoom in on a few key areas where there is especially high deforestation, like in the northeast. The semi-transparent colored blobs are protected areas. Some areas have surprisingly high deforestation.This is what is really at stake! The cutest creatures on the planet. We cannot let these unique animals and the habitats in which they live disappear.Cheirogaleus sibreei at Ranomafana National Park is one of the many diverse and critically endangered species found in this top priority protected area. C. sibreei is also found in the corridor forest north of Ranomafana which connects it to Fandriana, another top priority site. Habitat for Cheirogaleus sibreei in the Fandriana-Ranomafana corridor, north and outside the boundary of Ranomafana National Park. This corridor is technically protected. Slash-and-burn agriculture in habitat for Cheirogaleus sibreei in the corridor forest north of Ranomafana National Park.
|
AuthorJames P. Herrera Archives
January 2023
Categories |







 RSS Feed
RSS Feed
