|
Check out these videos featuring some of our DLC-SAVA Conservation projects and activities
lemur.duke.edu/protect/sava-conservation/sava-videos/ Huge thank you to everyone featured in the videos and especially Riccardo Morrales, videographer extraordinaire!
0 Comments
This is a teaser for the few of you who check my blog - thank you!
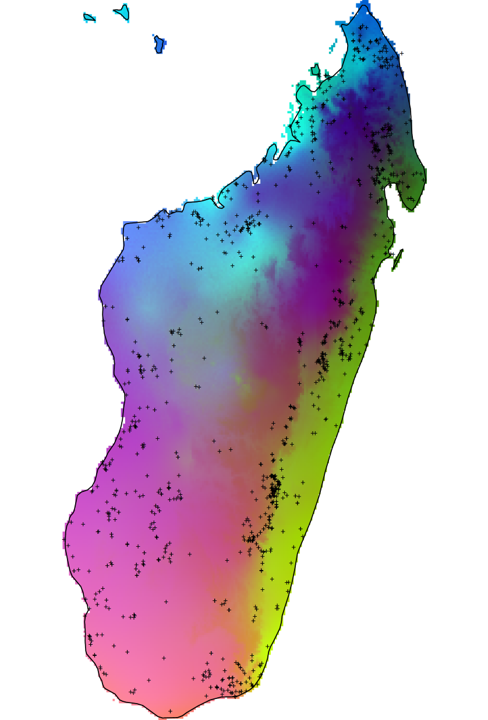
This beautiful rainbow of lemurdom illustrates the areas of endemism for lemur communities. Each dot is one of 1600+ occurrence records for 68 lemur species which I have compiled and very carefully curated over the last few months. Based on the relationship between the lemur occurrence records and the environment, this model predicts the spatial distribution of 'areas of endemism.' Areas with the same color are predicted to have a similar bunch of lemur species, and there are different species in regions with different colors. So each of these color clumps is a hotspot of unique lemurs not found elsewhere on the island. I'm investigating this to test how the environment and biogeography affect lemur diversity and distributions, and what it tells us about the way environment and history shape biodiversity distributions. There are a bunch more analyses on the way and I'm really excited about it because it's something I've been dreaming about since my first trip to Madagascar. I have always wanted to understand why lemurs are distributed where they are - is it the climate, or river barriers, or mountain barriers? There are literally dozens of hypotheses in the literature, and I'm using lemurs as a model for biogeographic evolution in Madagascar, and more generally. Stay tuned! shoot me a comment if you want to discuss methods, data, hypotheses! Here's some related reading: Niet0Lugilde et al, Elith et al Agroecology in SAVA region: DLC-SAVA Conservation interventions for sustainable development3/31/2020 James Herrera, Program Coordinator, Rabemanantsoa Thorien, Agriculture consultant, Charles Welch, Conservation Coordinator, Duke Lemur Center SAVA Conservation Farming practices around the world are changing to meet the needs of growing populations and adapting to climate change. The organic farming revolution has sparked a diverse subset of agricultural disciplines such as agroecology, regenerative and conservation agriculture, and permaculture. These practices seek to mimic and enhance natural processes to improve soil health, water management, and sustainable intensification. From a planetary health perspective, farming techniques that minimize environmental degradation will also greatly improve ecosystem and human health. In Madagascar, as in most of Africa, shifting agriculture is the leading cause of forest loss. Not only is shifting agriculture, or slash-and-burn, destructive for forest ecosystems, it also degrades the ecosystem services that nature provides for people, such as carbon sequestration, nutrient cycling, preserving soils, and water filtration / retention. Slash-and-burn agriculture is the most common way to prepare land before planting rice, manioc, or corn To help promote a shift from unsustainable slash-and-burn to more sustainable agroecology, the Duke Lemur Center SAVA Conservation began an agroecology training program. In collaboration with Peter Jensen of Terra Firma International, we taught skills in amending soils, composting, water management, and more. We described the first two workshops in this year’s newsletter. In November of 2019, 50 participants in two villages were trained in agroecology. Since then, DLC-SAVA agroecology technician Rabemanantsoa Thorien has continued to do monthly evaluations and consultations with the participants to track their progress and trouble shoot issues. Thus far, almost half of participants have already adopted the techniques we taught in their own home gardens and farms, and some have even begun teaching their neighbors! Here, we want to highlight some of the great successes we’re seeing. One participant, Jeannot from Ampahana, started his garden in December by creating “pocket ponds,” also called “keyholes,” to catch rainwater run-off and channel it through amended raised beds, full of compost, and covering the soil with mulch. He planted corn, beans, peanuts, zucchini, several other vegetables, and even rice in a polyculture, with biointensive seed spacing. By early February, Jeannot was already harvesting a crop and marveling at the rapid growth. We are extremely impressed, not only with his garden, but with his tree nursery where he’s preparing coffee and cacao to plant around systematically around his farm.  Left: Jeannot and friend Pierrot in December showing their newly planted and freshly mulched vegetable beds. Right: the garden less than three months later, with a lush polyculture of corn, peanuts, vegetables, lemon grass, and even rice! Another star participant is Dociline from Andasibe-Kobahina in the Andapa district. Dociline really embraced our principles of small, doable actions, making her garden close to home where she could experiment and manage daily with her new skillset. She created a garden of no more than 4X4 feet, in which she amended the soils, created a water catchment keyhole, and planted lima beans and peanuts. Although it didn’t look like much at first, by February her bean plants are taller than she is! Dociline is also a model for her neighbors and has already taught two of her neighbors to create gardens using the Terra Firma technique. Not only are these perennial plants producing nutritious beans, but they also encourage good microbes that fix nitrogen in the soil to improve fertility. Left: Dosiline days after creating her garden, a simple corner of her yard that was previously unused. Middle: she showed us how she had created her raised bed by amending the soil with rich compost she created, and is managing run-off water from her roof using the “keyhole” in the top right corner. Right: only 3 months later, her bean plants are prolific, growing up along a terrace of bamboo, and already flowering. These beans are nutritious, enrich the soil, and are perennial, producing food year after year. There are more than 20 additional farmers who have adopted the Terra Firma system. Some are comparing the results to garden plots using “traditional” techniques, and already remarking the difference in yields of corn and beans. As time goes on, and crops are rotated, it will be exciting to track their progress and share their successes! Thank you to Thorien who conducts follow up evaluations and consultations with our participants every month! Jean Marohavana from Manantenina shows off his flourishing ginger and corn plantation, which is also surrounded by diverse vegetable crops. These soils were bare and rock-hard when we conducted the workshop in November. Jean has since amended the soils, optimized rainwater run-off, and is reaping the rewards of his labor. Ginger is an important cash crop in the region. Rabemanantsoa Thorien, DLC-SAVA Agroecology tech, right, with farmer Paul Marohavana from Manantenina on left
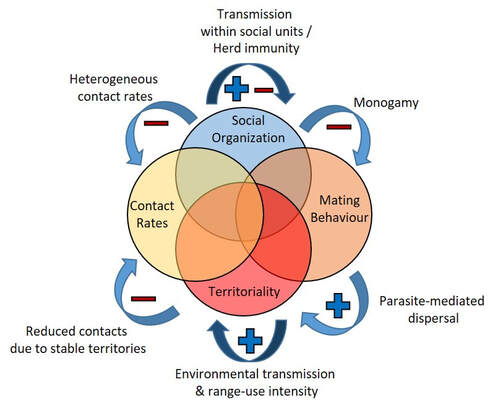
The biodiversity on earth is spectacular, with over a million species known to science, and at least 10 times as many species still to be discovered. Biodiversity is amazing in its own right, but is also vital to human well-being, providing resources and services for people, including building materials, pharmaceuticals, clean water and air, as well as recreational areas. Despite the value of biodiversity, many species are threatened with extinction by human activities. Deforestation, overharvesting, and climate change all put species at risk of extinction. Disease increases the vulnerability of species, and when combined with other factors, can wipe out whole populations. Of the species that have already gone extinct, disease was a likely cause for 4% of them. Parasites that cause diseases are also important parts of ecosystems, but when species are threatened by habitat loss and other factors, the natural balance between animals and their parasites can be disrupted, leading to epidemics that can negatively impact threatened populations. In a new article published this month, my colleague, Dr. Charles Nunn, and I reviewed the literature on the links between disease, animal behavior, and conservation. This article is part of a special theme issue of the Philosophical Transactions of the Royal Society, specifically focused on how important understanding behavior is for conservation. We detail many theories and examples of the interplay between animal behavior, disease, and protecting biodiversity. For example, sociality in animals impacts their disease risks. Animals that live in larger groups often have more parasites than those that are solitary or live in small groups. This pattern is not universal, though, and the benefits of sociality likely outweigh the costs of disease. Specifically, living in groups allows wolves to have more successful hunts than those that live alone, despite the risk of transmitting mange among individuals. Living in groups also facilitates ‘herd immunity,’ a principle similar to vaccination in which exposure to disease-causing parasites allows group-living individuals to develop antibodies that make them resistant to the parasites. These behaviors have important implications for conservation. One common method for managing diseases in populations is to remove sick individuals, either by quarantining or culling herds. However, such management practices can have adverse effects, especially when they disrupt natural territories and lead to colonization of managed areas by animals from other places, which can bring diseases with them. Careful management can incorporate aspects of behavior to identify the highest priority animals to treat, and combining vaccination, contraception, and culling can be more effective than any one strategy alone. While the examples above illustrate how behavior affects disease, the opposite is also true – diseases affect behavior. Some of the most fascinating examples include parasites that can directly manipulate host behavior to facilitate their own transmission. Rabies is a classic example, caused by a virus that infects parts of the central nervous system, including the brain, increasing aggressive behavior and salivation. Infected animals bite others, leading to disease transmission. Rabies can have severe impacts for endangered species, such as an epidemic that devastated Ethiopian wolf populations. Toxoplasma is a parasite with multiple hosts in its life cycle. It initially infects small mammals and leads to a decrease in their fear responses to predator cues. For example, infected rodents show no fear to cat urine, and infection leads rodents to be preyed on by cats more often, facilitating the transmission to the cat. These phenomena are important for managing threatened species because the transmission of these parasites can have negative impacts on both predator and prey populations. Policies to manage threatened predators must also evaluate the prevalence of these parasites in the hosts, and multispecies parasites that negatively impact endangered species may persist because they also infect more common hosts. The interplay of behavior, disease, and conservation is especially important now as species are threatened by habitat loss and climate change, which are negatively impacting the geographic ranges and population sizes of many species. To effectively manage threatened species in the future, it will be important to create policies that integrate aspects of how behavior affects disease and vice-versa, and interdisciplinary research will be especially relevant in this context.
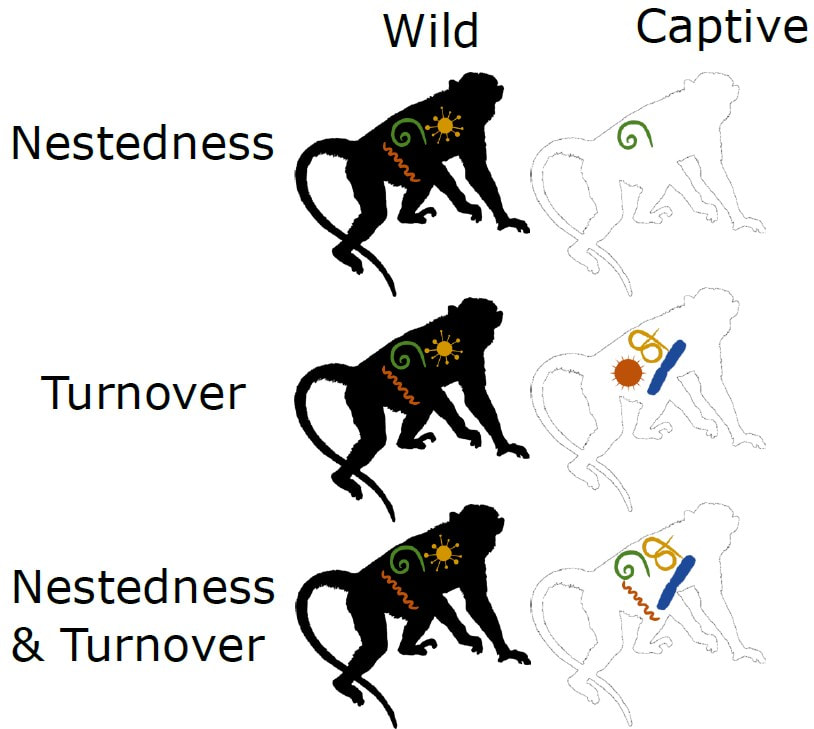
Parasites include many kinds of bacteria, viruses, and worms, among others. These are organisms that live on or in another organism (the host), from which they get their resources, at some cost to the host. Parasites are all around us, and people are known to get at least 1400 parasites from other animals! Many parasites can cause disease and are harmful. They aren’t all bad, though, because they play important roles in ecosystems, similar to predators. Primates include lemurs, tarsiers, monkeys, apes, and people. Because we’re so closely related to wild primates, we can get a lot of the same parasites. One of the most famous includes the human immunodeficiency virus (HIV), which came from SIV (simian immunodeficiency virus). There are lots of parasites that infect many mammals – they are generalists that can infect different host species. Other parasites are specialized, and are only found in a single host species. These complex patterns make it hard to predict which parasites can be shared among species or in different environments. Primates are common in captivity for many reasons. They are a favorite in zoos and sanctuaries, and they are used in biomedical research – for better or worse. While in these captive settings, primates get medical treatment, including quarantines before being introduced to new groups, frequent testing for parasites, deworming and antibiotics, as well as high standards for hygiene. Given these conditions, we predicted that captive primates have fewer parasites than their wild counterparts. In a study published recently in the American Journal of Primatology, we tested this prediction to find out if parasites are lost in captivity. We compared the presence of almost 600 parasites known from 23 primate species for which data are available in the wild and in captivity in the published literature. With these paired comparisons, we tested if captive primates have fewer parasites than the same species in the wild. Counter to our predictions, there was no overall difference in parasite species richness between wild and captive primates. This was surprising because we expected that the sterilized captive environment and access to veterinary care would eliminate many wild parasites in captivity. We also compared which parasite species are found in primates in the two settings, and how the parasites found in each setting differed. We found that the parasite communities were significantly different between the wild and captive conditions. Primates in the wild have diverse parasite communities that include those with vectors and intermediate hosts (e.g., mosquito- or tick-borne, or parasites that have different life stages in different host species). In contrast, primates in captivity have mostly parasites that are transmitted through the environment, especially those spread through feces, and via close-contact. Further, captive primate species have many parasites that are known to infect people, livestock, and other domesticated animals. Similar results have been shown for the microorganisms in the guts of primates; captivity makes the community of gut bacteria of captive primates more similar to that of people than to the wild gut bacteria, with important implications for health.
Don’t be afraid of getting sick from primates at the zoo. In our study, we did not actually test individual zoo animals and find parasites; we found published studies from a range of facilities. I suggest you do not touch captive animals or their enclosures, and you will have very little risk of getting a parasite from zoo animals. Actually, you can be more dangerous to the primates than they are to you in terms of diseases. Primates can get sick from our diseases if you try to feed or touch them. Also, people who maintain captive primates should be aware of the parasites to which they are exposed, and protect themselves when in close proximity to primates, and their environments. The same is true in the opposite direction: captive and wild primates are at risk of getting parasites from people that can be deadly – this is a risk for the captive colony, as well as wild populations. This is another great reason why primates are not pets. Primates can get sick from our germs, and we can get sick from theirs. In our new article, my colleagues and I found strong relationships between the distribution and abundance of primates from Madagascar and the trees from which they eat fruits, leaves, and flowers. We then used those relationships to make estimates of the total population sizes for endangered species to better understand their conservation status. The paper, “Estimating the population size of lemurs based on their mutualist food trees”, was just published in the Journal of Biogeography. Lemurs are the endemic primates of Madagascar, found nowhere else on earth. Because of their long isolation and unique ecologies, more than 100 lemur species have evolved. This amazing diversity of species is threatened by the loss of habitat in Madagascar, as well as hunting. Recent efforts have shown that over 90% of lemurs are threatened with extinction. It is critically important to estimate the total population size of threatened species to monitor their potential decline in the future. Most lemurs are herbivores, and some rely heavily on a few tree species for food. Many lemurs eat fruits and spread their seeds, making them important for healthy ecosystems. Previous research on many groups of plants and animals has illustrated the importance of interactions among species in determining the geographic distributions and abundances of species. For example, some butterfly species need specific host plants, and so the butterfly cannot exist in a place if the host plant does not exist, even if the climate is appropriate. In our study, we use the distribution of 14 lemur food trees to map the distribution of 19 lemur species and estimate their abundance. Using new data from field observations of endangered lemur species and statistical methods, we created estimates of the total range and population sizes of lemurs. We found that the geographic range of all 19 lemurs and the abundance of most lemurs is correlated with the distribution of the food trees. For all lemurs, the lemur range was strongly correlated with the range of their food trees, more so than with climate alone. Our estimates of abundance also show that for most species, the population sizes are greater than 10,000 individuals, which is the size below which the species is considered threatened with extinction. For example, the ring-tailed lemur (Lemur catta) may have a total population size of over 500,000 individuals! This is great news because recent press by BBC and IFLScience have suggested lemurs may be 'doomed' with extinction. Ring-tailed lemur in Isalo National Park. Ring-tailed lemurs may be abundant, but collecting animals for meat and the pet trade still threaten fragmented populations. In contrast, the sportive lemurs in the southeast (genus Lepilemur) are relatively rare, with a high estimate of 7,000 individuals. The black-and-white ruffed lemur (genus Varecia) is particularly vulnerable, with the lowest estimates of approximately 1,400 individuals. Sibree's dwarf lemur (Cheirogaleus sibreei) may have less than 2,000 individuals in the southeast, though the species's range includes sites in central and northeastern Madagascar as well. Some lemurs had population sizes greater than 10,000 individuals, but are still at a high risk of extinction due to unsustainable hunting and deforestation. In another paper recently published in Conservation Biology, my colleague Dr. Cara Brook showed that one species, the indri, may go extinct because of hunting in some areas. The sportive and black-and-white ruffed lemurs have fewer than 10,000 individuals in the wild Sibree's dwarf lemur is only found on high mountains  The Indri is threatened by hunting pressure in the northeast For several species, we were able to compare our estimates of recent population size to past estimates from the year 2000. At least two species seem to have declined in population size dramatically. While approximately 100,000 red-fronted brown lemur (Eulemur rufifrons) were estimated to exist in the southeast around the year 2000, we estimated that only 15,000-20,000 individuals occurred in 2014. Another species, the gentle bamboo lemur (Hapalemur griseus) from the southeast was estimated to have less than 20,000 individuals in the year 2000 while we estimate there to be approximately 10,000 in 2014. These trends are related to habitat loss as well as the new methods we used to estimate the population sizes. The red-fronted brown and gentle bamboo lemurs have much smaller population sizes than estimated in the year 2000 These results are significant because many lemur species are important for spreading seeds and as food for predators, and they are a unique branch on the primate family tree. If they go extinct, we will lose those ecosystem functions and evolutionary history. The Malagasy government has taken action to protect the most valuable and diverse habitats remaining in Madagascar. Malagasy and international conservationists and researchers work closely with the government, educational system, and nongovernment organizations to turn research results into conservation action. More effort is needed because Madagascar is an extremely impoverished country with a high population growth rate and a high demand for natural resources.
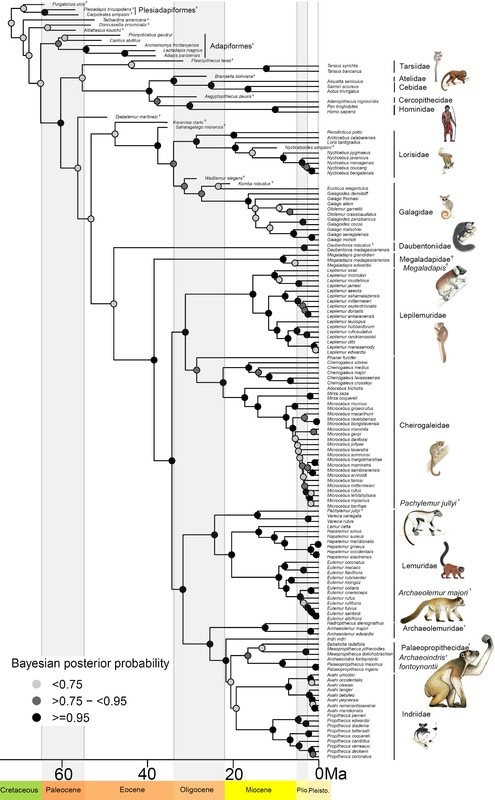
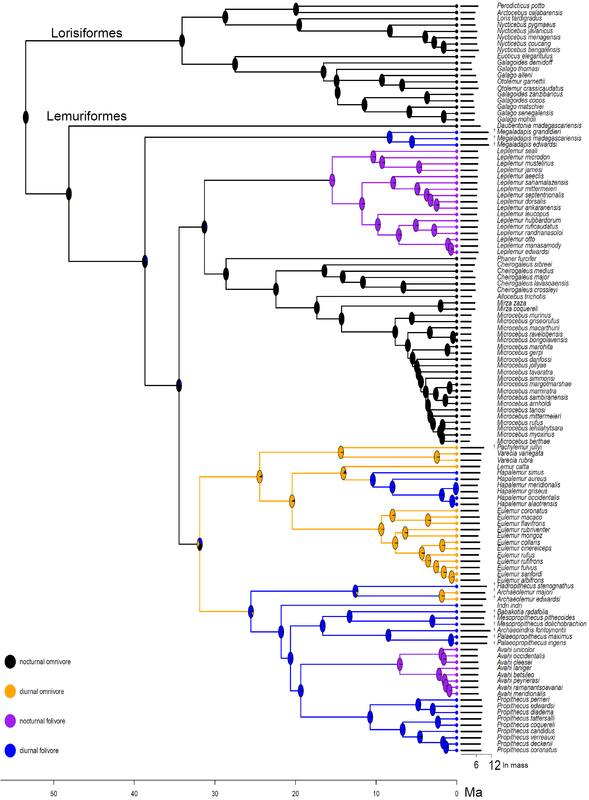
In a new article available online at the Royal Society Open Science, I tested several predictions of adaptive radiation theory for lemurs of Madagascar. This charismatic group has long been suggested to be the result of adaptive radiation, but my results suggest the long evolutionary history of lemurs may not fit all the predictions. Adaptive radiation is a key concept in evolutionary biology, with a fantastic diversity of species arising to fill every conceivable niche. Famous examples include Darwin’s finches of the Galapagos, lizards of the Caribbean, and cichlid fishes in Africa. Madagascar, an island nation off the coast of Africa, is a biodiversity hotspot, with thousands of species found nowhere else on earth. The primates – lemurs – have long been suggested to be an example of adaptive radiation, with around 100 species alive today that have unique and diverse niches. The theory of adaptive radiation makes several explicit predictions, which have not yet been tested for lemurs. For example, evolution of morphological adaptations to fill unique niches is an important part of adaptive radiation theory. Do lemurs fit these predictions? In a new article accepted in the Royal Society Open Science, I tested multiple predictions about the rate that species diversified, and how their body size adapted to different niches of diet and activity pattern. I also tested the possibility that a mass extinction event in the past, 34 million years ago, may have changed the rate of speciation and extinction. While theory predicts that speciation rates should be highest early in the evolution of an adaptive radiation and slow down towards the present, lemur speciation rates were either constant through time, or may have even increased slightly over time. The rate of body size evolution fit predictions better than speciation rates; the major groups of lemurs seem to have differentiated into unique niches with different optimal body sizes. Lemurs that eat leaves and are active during the day have larger body size than day-active fruit eating lemurs. There are three unique groups of small-bodied lemurs that have independently evolved adaptations for eating leaves. Additionally, there are small nocturnal species adapted for eating fruits, nectar, and insects. This diversity of niches and associated body size evolution suggests that lemurs may have evolved via adaptive radiation, despite the contrary results from speciation rates. While there was no evidence that a mass extinction event disrupted speciation dynamics in the deep past, 17 species of giant lemurs have gone extinct within the last 2,000 years, not long after the arrival of humans around 4,000 years ago. The giant lemurs were unlike most alive today, with one species as large as a silverback gorilla. The loss of these unique species represents a significant change in Madagascar ecology and evolutionary history. This study provided explicit tests of the adaptive radiation theory for lemurs, with mixed results suggesting that body size and niche evolution fit expectations, but speciation rates may tell a different tale. Lemur phenotypes - or their morphology, ecology, and behavior - are so diverse, they fit the prediction of adaptive radiation. While some species are small, nocturnal omnivores, including the smallest primates, the mouse lemurs, other lemurs are larger, diurnal and eat leaves. Some species are even specialized to eat bamboo with enough cyanide to kill people! The largest living lemur, the 6-10kg (12-20lbs) Indri, is tiny compared to the giant lemurs that went extinct just 2,000 years ago, which were as big as gorillas - up to 150kg, or 300lbs! Phylogeny of lemurs and other primates, illustrating living and extinct species. Figure from Herrera and Davalos 2016, Syst. Biol. Illustrations courtesy of S. Nash, used with permission. Strepsirrhine phylogeny illustrating the distribution of body mass (black bars next to tips, scale at the bottom, natural log transformed) and adaptive niches of diet and activity pattern (colored circles next to tips). The evolution of niches was estimated at internal nodes (colored pie charts on nodes representing the proportional probability of each state) and the best estimate at each node was assigned to the descendant lineages (colored branches). This tree was used to test how body mass evolved at different rates in different niches.
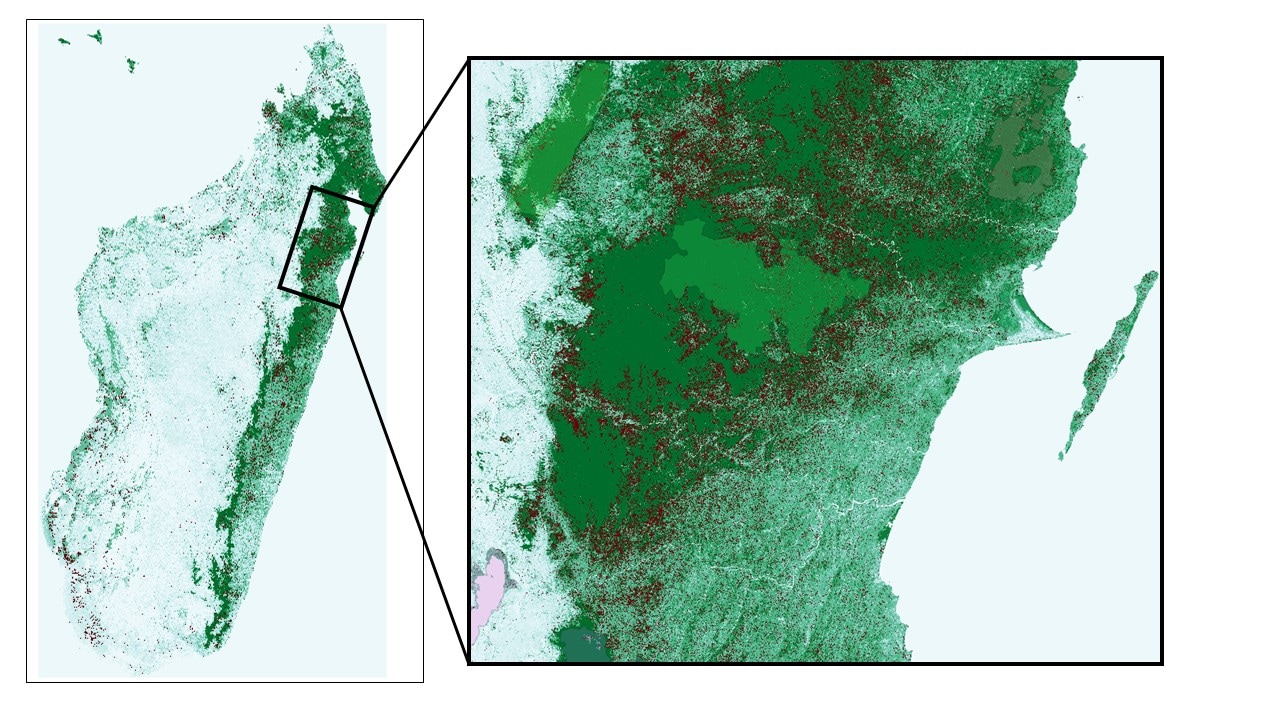
My latest paper on lemur conservation priorities just came out in the journal of Biological Conservation. In it, I discuss a new priority scheme for 100 protected areas that have high lemur diversity in terms of threatened species, functional traits of species, their evolutionary history, as well as the quality and threat to their habitat. This multidimensional approach allowed me to rank protected areas based on a diversity of measures. Biodiversity worldwide is threatened with extinction due to human activities. Primates, the group including monkeys, apes, and people, are vulnerable to habitat loss, hunting, and future climate change. 44% of primate species are considered threatened by the International Union for the Conservation of Nature and Natural Resources (IUCN). The lemurs of Madagascar, found nowhere else, are the most endangered primate group, with 93% threatened due rapid habitat loss, as well as hunting in some areas. There has been a tremendous effort to conserve lemurs and the habitats they thrive in, including the creation of over 100 parks and reserves (protected areas, PAs) to protect montane rainforests, tropical dry forest, and arid spiny forest. Madagascar is an impoverished nation, however, with limited funds to maintain PAs. In 2013, conservation priorities for PAs were set based on the number of species in each PA and how endangered they are in the Lemur Action Plan. In a new study, I propose a new priority ranking for 100 PAs based on their lemur diversity and habitat quality. Assessing diversity is difficult, though – how should we calculate the value of a PA for lemurs? I quantified diversity in terms of 1) the number of species, 2) how endangered those species are, 3) how diverse the functional traits of species are, 4) how much evolutionary history species represent, and 5) how diverse and productive the habitats are. To measure the level of anthropogenic threat to these PAs, I calculated the rates of deforestation using data from Hansen et al. 2013 (data found here). I found that the priority ranking of PAs differed depending on the measure used, leading me to create a summary metric that captures the importance of each PA on multiple levels of diversity and threat. Surprisingly, I found that deforestation inside of PAs was high; up to 31% forest loss between the years 2000 and 2014, totaling ~3,000 km2, which is 50 times the size of Manhattan. The highest deforestation occurred in PAs with high species richness and functional trait diversity. I ranked PAs to suggest the priorities for more intensive future conservation efforts. The top PA is a corridor in the southeast that links the Fandriana National Park to the Marolambo forest south of it, and has only recently been declared. Corridor forests are especially important as a means for individuals to disperse among populations and aid gene flow, as well as a means of repopulation if some populations suffer declines. Based on phylogenetic diversity - the evolutionary distance separating the species in the park - some internationally recognized PAs were found to be priorities, such as one UNESCO world heritage site (Tsingy de Bemaraha). There are also several highly threatened environments with high functional diversity, especially in the arid southwest like Beza Mahafaly. With these new priority rankings, scientists can collaborate to focus their efforts on the most threatened and diverse PAs. Efforts should also be maintained in successful PAs, which have had limited deforestation. These sites have created partnerships with local stakeholders to collectively protected natural habitats, such as Ranomafana and Analamazaotra. The full list of PAs, and their ranks based on each metric can be downloaded here. While there are many impediments to conservation in Madagascar, especially high deforestation for agriculture, there are many success stories which provide a framework for conserving species in the future. Much remains to be learned about conservation priorities in Madagascar. It is especially important to quantify the importance of these PAs for the people of Madagascar. PAs are important at the national level because of the revenue generated by tourism. They significantly impact the lives of local populations surrounding them, who depend on the land for subsistence, like farming and building materials, as well as for ecosystem services such as clean water. Parks generate revenue for local populations through tourism and research, but often the economic loss of restricted access to land is not offset. Lastly, some forests have spiritual value, because some ethnic groups bury their dead in tombs within forests. Therefore, nature conservation must also determine the value of PAs for people. Another way this study can be improved is to enumerate these diversity data for multiple taxonomic groups. These data are accumulating with many field projects collecting data on diverse groups such as birds, small mammals, bats, amphibians, reptiles, and plants. Combining these new occurrence records for diverse groups will allow a more multifaceted approach to conservation in Madagascar. To summarize, this new paper reports conservation priorities based on the sum of lemur diversity, habitat quality, and deforestation. Rapid loss of habitat is threatening important biological systems that also provide crucial services for people. I hope that the results of this study will be well received by the community and can help guide future conservation efforts. Below are figures illustrating the extant of forest in the year 2000 (green gradient) and the forest loss is in red. I also zoom in on a few key areas where there is especially high deforestation, like in the northeast. The semi-transparent colored blobs are protected areas. Some areas have surprisingly high deforestation.This is what is really at stake! The cutest creatures on the planet. We cannot let these unique animals and the habitats in which they live disappear.Cheirogaleus sibreei at Ranomafana National Park is one of the many diverse and critically endangered species found in this top priority protected area. C. sibreei is also found in the corridor forest north of Ranomafana which connects it to Fandriana, another top priority site. Habitat for Cheirogaleus sibreei in the Fandriana-Ranomafana corridor, north and outside the boundary of Ranomafana National Park. This corridor is technically protected. Slash-and-burn agriculture in habitat for Cheirogaleus sibreei in the corridor forest north of Ranomafana National Park.
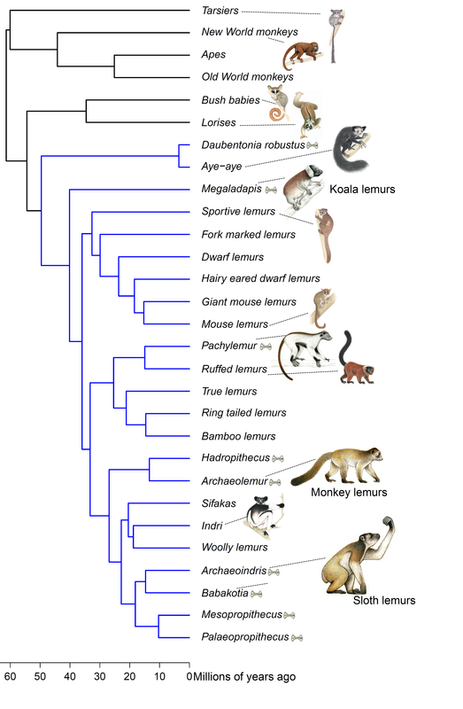
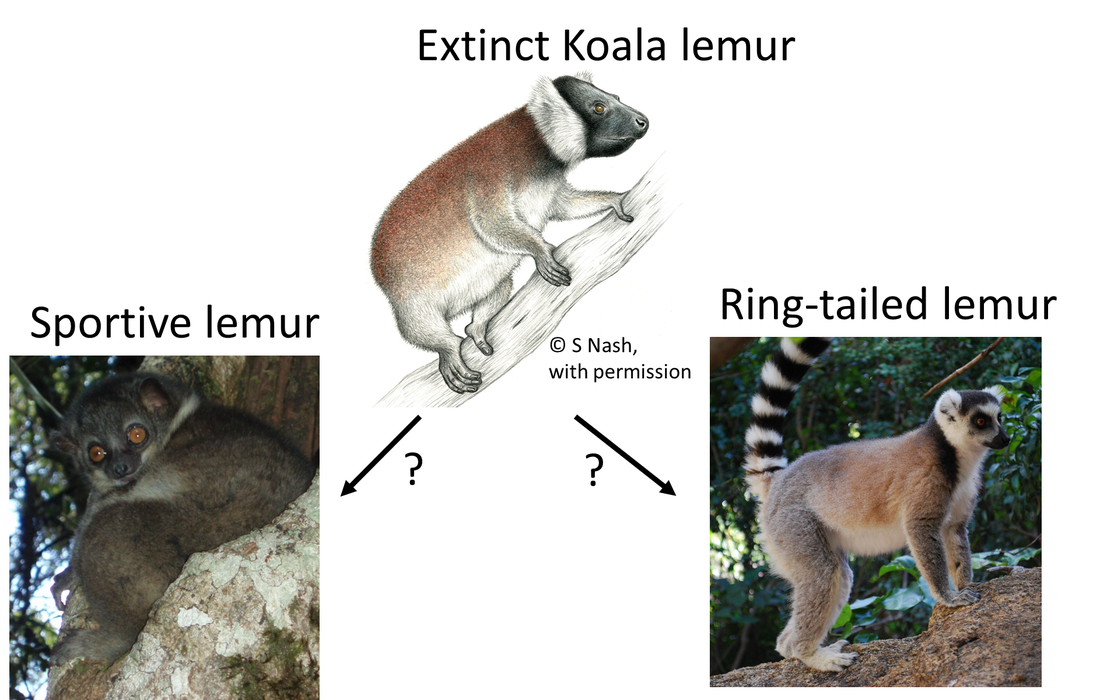 We humans —along with apes, monkeys, and lemurs— are primates, and exploring primate evolution helps us understand where we came from. Indigenous to Madagascar, lemurs form a unique branch of the primate family tree. But there is one important obstacle to understanding where lemurs came from and how they evolved– there are no ancient fossil lemurs. This void makes it difficult to track the stages of evolution often observed in transitional fossil forms. Making matters more confusing, but fascinating, there are remains of lemurs extinct around 2,000 years old. Those lemurs were radically different from the ones alive today. Many of the extinct lemurs were giants, with some weighing as much as gorillas! Placing them in the tree with living species has been difficult because of their unique features. To understand the factors that may place species at risk of extinction, it is important to compare the traits the extinct species had to those of living species. For my Ph.D. research, the first step was to do just that. With my committee member and collaborator, Stony Brook University Professor Dr. Liliana M. Dávalos, I studied the evolutionary history of lemurs in detail with support from the National Science Foundation. The findings are out this week in the journal Systematic Biology. For the first time since the early 1990s, I combined data from living and extinct species with cutting-edge techniques to place all lemurs in a single tree, providing the most complete tree of lemur evolution yet. Surprisingly, the new phylogeny finds some of the subfossil species were unique branches with no living close relatives. The giant, extinct koala lemur (Megaladapis) had short limbs and long finger and toe bones, which may have allowed it to cling to branches, much as living koalas do. Megaladapis also had an extremely long snout with no upper front teeth, and may have even had long dexterous lips for manipulating food. The koala lemur was thought to be closely related to either of two living groups: the sportive lemurs (Lepilemur), or ‘true’ lemurs, like the ring-tailed lemur (Lemuridae). Because of the unique features with few similarities to living species, placing them in the lemur tree based on skeletal traits has been difficult. Fragments of ancient DNA had suggested a close link between koala and true lemurs. By combining genetic and anatomical data, the new tree finds the koala lemur was a completely extinct, unique branch of the lemur tree. Other fossil groups are more closely related to living species, but the fossil groups were giant compared to living species. While the largest living lemur is about 22 pounds, the largest and closely related extinct lemurs may have weighed over 300 pounds! Large body size is one important component of extinction risk, and so by understanding the evolution of body size, we can better understand the factors threatening lemurs today. The implications for the future of lemurs are substantial; 95% of living lemurs are threatened with extinction. With all living and extinct lemurs together in the same tree, we can begin to understand how evolution and extinction have worked in the past, and project risks into the future. Another way the study helps us understand primate evolution is by illuminating how geography has contributed to splitting off species. Madagascar has been an island separate from all other landmasses for at least 90 million years, long before primates first appeared. As in previous analyses of molecular clocks, the new study suggest lemurs first evolved ~50-60 million years ago. This means the ancestor of lemurs likely arrived on Madagascar by dispersal – perhaps washed from Africa to Madagascar floating on vegetation, or with some small stepping-stone islands to help along the way. A surprising new twist arises from the new lemur family tree: could lemurs have dispersed from Madagascar back to Africa? In some analyses, there was evidence a fossil primate found on Africa 34 million years ago (Plesiopithecus) was most closely related to the aye-aye, the first lemur to diverge from other species. If confirmed, this evolutionary pattern means the aye-aye and the ancestor of all other lemurs dispersed from Africa to Madagascar separately, or the ancestor of all lemurs arrived on Madagascar and this unique branch dispersed back to Africa. This finding warrants an intense focus on the similarities between the aye-aye and the African fossil Plesiopithecus to determine whether they are the result of common ancestry, or if this link is an artifact of similar ecological pressures leading to similar traits. With this new evolutionary tree, we have a much better understanding of the evolutionary dynamics of primates and how evolution shaped the primates of Madagascar over the last 50 – 60 million years. We can now compare the factors associated with the extinction of all subfossil lemurs, and predict how living lemurs may fare in the future. This information is crucial if we hope to prevent further extinctions of these distant human relatives. Check out the Huffington Post's piece on this article And Forbes too! Illustrations copyright S. Nash, used with permission
The first chapter of my dissertation to be published is now out in the Journal of Animal Ecology, detailing a close connection between the diversity of primates and the abundance of their food tree resources. Lemurs are primates endemic to Madagascar – they are indigenous and found nowhere else on earth. They are also highly threatened with extinction; 95% of lemurs are considered at risk of extinction. The primary threats to lemur persistence are habitat loss due to deforestation for agriculture, cattle grazing, logging, and mining, as well as hunting for bush meat. The new study illustrates the tight links between lemur diversity and ecosystem health.
In case this is your first time on my page, my name is James Herrera, and I am a researcher at the American Museum of Natural History. I conducted the study as a Ph.D. student at Stony Brook University. I spent two years trekking across rainforests in southeast Madagascar to count lemur species and determine their abundance in relation to habitat characteristics such as human disturbance, altitude, and tree community composition. I surveyed five different habitats with teams of Malagasy field assistants and graduate students. The study was conducted in Ranomafana National Park and an adjacent forest corridor that connects Ranomafana to the Fandriana Park to the north. Some habitats were low altitude pristine rainforests like green cathedrals of massive, broad canopy trees. Other habitats were high mountain peaks of granite, festooned with forest clinging to rocks. I also surveyed heavily disturbed sites, some of which had been logged or cultivated in the past and subsequently grew back, while others were actively being logged and mined for gold around his camp site. This variety of habitat types allowed me to compare the primate communities in relation to variation in habitat features. The study revealed the overwhelming effect of food tree abundance on lemur diversity. While some small nocturnal lemurs eat insects, many lemurs are vegetarians, and most specialize on the fruits or leaves of just a few key tree species to obtain most of their diet. Collaborating with Malagasy botanists, I enumerated the food trees in each habitat compared to non-food trees, and found the relative abundance of preferred food trees was most closely tied to lemur diversity. Where food trees were most abundant, the lemur communities were dominated by a few large-bodied species that form large groups, while small species with small group size were rare or absent. When food trees became scarce, those previously dominant large-bodied species became rare or absent and other species increased in abundance. This result illustrates how resource limitation affects the abundance and diversity of lemurs. This finding is especially pertinent for healthy ecosystem functioning. The fruit-eating lemurs are important seed dispersers; when lemurs eat the fruits, they pass the seeds whole which germinate much better than seeds not passed by lemurs. Different lemurs specialize on different trees and disperse the seeds far from the parent trees, like gardeners sowing the future forests. Without these lemurs, the trees may not be able to reproduce as efficiently, and so lemur extinctions could have cascading effects on ecosystems. Surprisingly, human disturbance did not have a strong effect on lemur diversity, possibly because of the indirect effects on food tree abundance. For example, while some heavily degraded sites had low food tree abundance, so did high altitude pristine forests. The study illustrates that some lemur species may be resilient to habitat disturbance, especially those small-bodied species that have small group sizes and can be sustained with lower resource abundance. The result is important for understanding how entire lemur communities will respond to future habitat loss, which is especially important because of the ongoing degradation of natural habitats. While I surveyed corridor forests that connect two national parks, migrants from far-away cities were cutting forest, damming rivers and panning for gold. The recent surge is especially troubling because it attracts bandits who rob gold miners and threaten the safety of rural villages. Further, many of the miners are immigrants from cities, not the local land owners with rights to the forest resources. Protective action is needed to suppress the burgeoning deforestation, and this corridor is especially important to maintain safe passageways for lemurs to move among parks and maintain healthy stable populations. This study was funded by the National Science Foundation, Margot Marsh Biodiversity Foundation, Conservation International, Explorer’s Club, Leakey Foundation, Primate Conservation, Inc., Rufford Small Grants Foundation, International Primatological Society, American Society of Primatologists, La Conservatoire pour la Protecion des Primates, and the Turner Fellowship while at Stony Brook University, as well as a Gerstner Scholarship and Postdoctoral Research Fellowship from the Richard Gilder Graduate School, American Museum of Natural History. I also acknowledge the help of over 30 Malagasy research assistants, especially Tongasoa Lydia, a Malagasy Ph.D. student who worked with me during this study. |
AuthorJames P. Herrera Archives
January 2023
Categories |















 RSS Feed
RSS Feed
